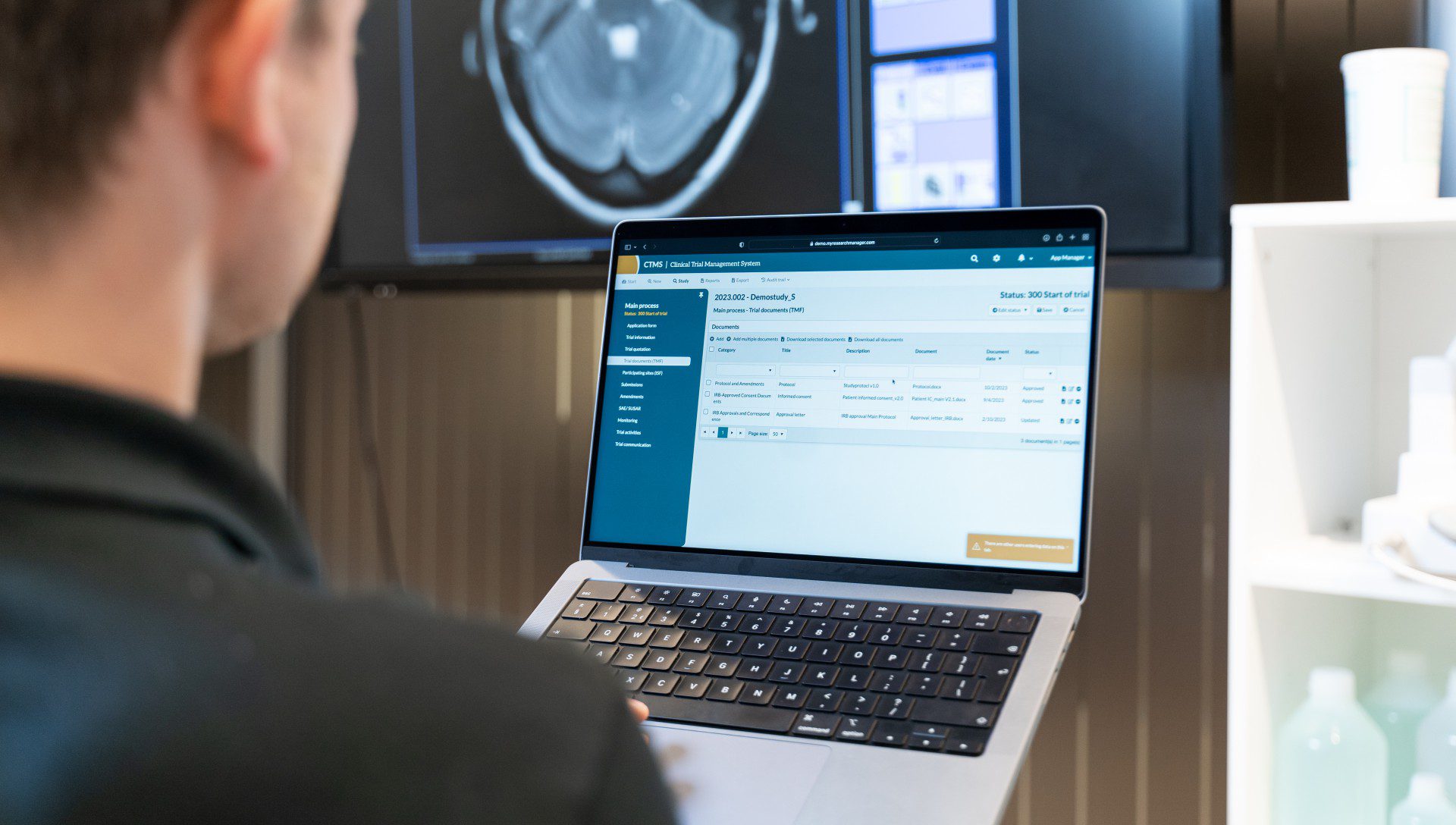
Our experts assist in setting up the Clinical Research Suite
Professional Services
Elevate clinical research to a higher level. Our Professional Services complement the Clinical Research Suite and provide support in setting up, configuring, and executing the clinical research process. These tools, features, and personalized support combine our years of experience with a focus on powerful and modern technology. Utilize ResearchManager Professional Services for an optimal, reliable, secure, and sustainable research process.

Study Management
A successful study begins with a well-designed protocol and well-organized study processes. With our Study Management, we provide support in every phase of the research process. We focus on planning, managing, and coordinating the study, ensuring that it stays on track and within budget. Did you know that Site Management is also crucial? Effective supervision at research sites, along with maintaining strict compliance with regulations and quality standards, is essential. Lastly, Study Management provides an efficient planning tool. Study Management is the foundation for a high-quality and successful research endeavor.
Contact us
Regulatory
Now that we have the study on track, it’s time for the next phase: Preparation and Regulation. This phase is crucial for a reliable study. We support researchers with Regulatory Submissions, ensuring that documentation is submitted to relevant authorities in a timely and accurate manner. With Legal Representation, the study complies with regulations and provides legal assurance, while Investigator Selection & Site Qualification assists in carefully selecting the research team and suitable locations. Finally, Investigator Meetings ensure that researchers are well-informed and stay compliant with the prescribed study protocols.
Contact us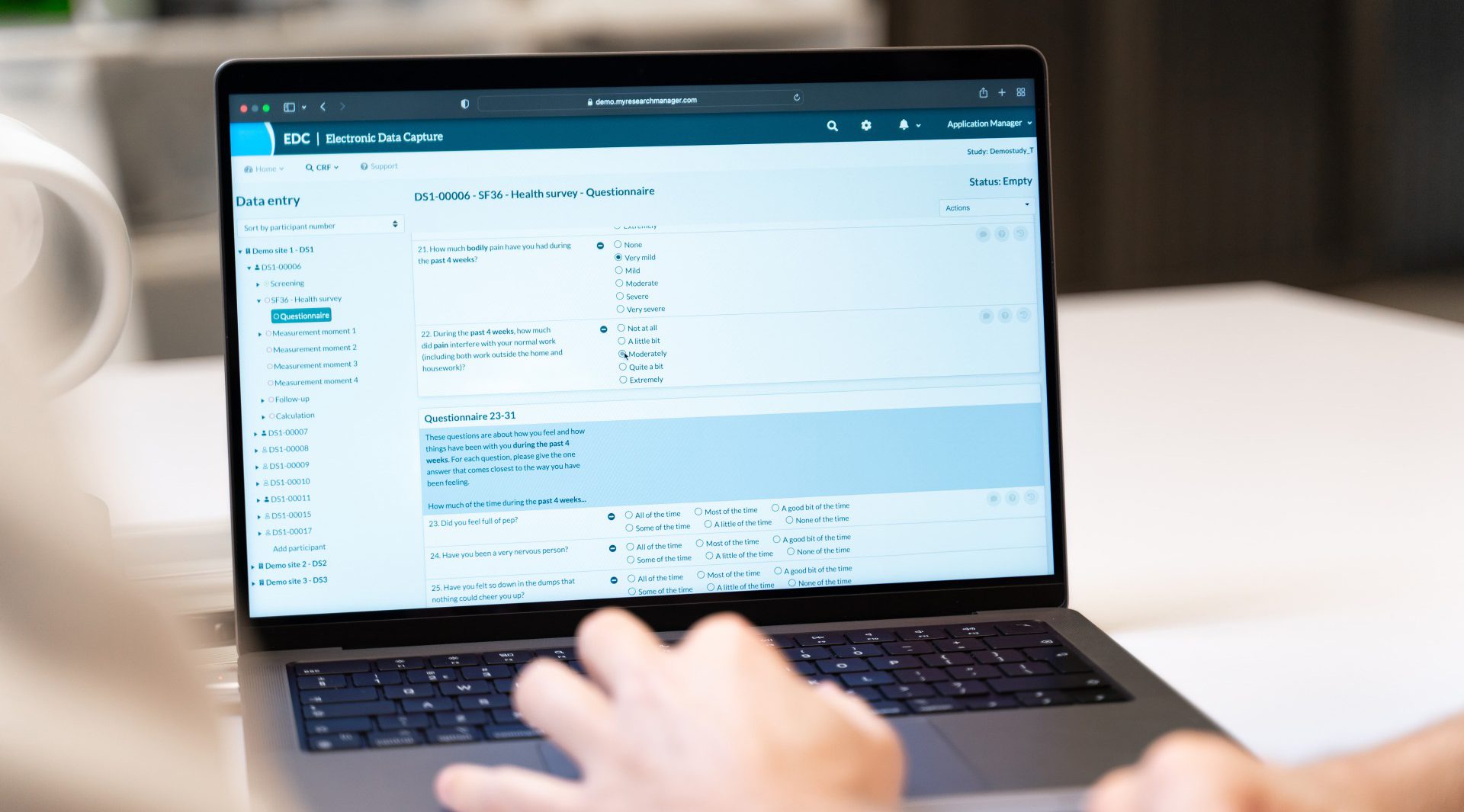
Data Management
Data management is crucial in this phase, requiring accurate data management and reporting. Our services in Data Management and Statistics ensure consistent and reliable data collection. Analysis and reporting are also indispensable elements. Our expertise in Medical Writing is essential for drafting required documentation, while Safety Reporting ensures reporting to relevant authorities.
Contact us
Monitoring
Now that all preparations have been made, it’s time to set up Monitoring & Oversight. With these tools, we ensure continuous and accurate data control, guaranteeing that data is collected correctly. We also conduct (on-site) QA audits, both on location and remotely, to confirm that all activities within the research process meet quality standards and do not jeopardize the integrity of the study. With Surgery Support Service, we ensure compliance with study protocols during surgical procedures conducted for research purposes.
Contact us
Reports
After data and information collection and control, we handle Reporting & Compliance. Our services for Data Management and Statistics provide reliable data collection, thorough analysis, and clear reporting. These reports meet legal requirements and are prepared by experts in clinical research, combining our years of experience in clinical research with the modern Clinical Research Suite.
Contact us